NONWOVEN fabrics POLYESTER Fibers
(Educational Research)Ramaiah Kotra and Xiao Gao
In 1996, 24.1 million metric tons of manmade Fibers were produced worldwide. This was 4% more than in the previous year. The main volume gain took place in production of PET Fibers (PET filament 9%, PET staple 4%) [1]. The primary drive for this growth is demand for fiber and container resin. Seventy five percent of the entire PET production is directed toward fiber manufacturing. Hoechst, DuPont and Eastman are the three world largest polyester producers. Additional current U.S. Polyester fiber Producers are: Acordis Industrial Fibers, Inc.; AlliedSignal Inc; Cookson Fibers, Inc.; Kg5A; Intercontinental Polymers, Inc., Martin Color-Fi. Nan Ya Plastics Corp., Wellman, Inc. [24] Dramatic growth in PET fiber production is foreseen in Asia in the near future [22].
The cost of polyester, with the combination of its superior strength and resilience, is lower than that of rayon. Polyester Fibers are hydrophobic, which is desirable for lightweight facing fabrics used in the disposable industry. They provide a perceptible dry feel on the facing, even when the inner absorbent media is saturated. As new methods of processing and bonding of PET are developed, rayon is being replaced by polyester on the market. According to David Harrison [2], 49% of the total nonwovens market share in the USA belongs to polyester staple, reaching 291 million pounds in 1996 and ranking number one among all kind of fiber supplies. If one were to assume that the filament fiber consumption is half of that of staple, the total PET consumption in the USA nonwovens industry alone would be over 450 million pounds. Even if the estimate is not be completely accurate, polyester has become the most widely used polymer in the nonwovens industry since 1995. The next most popular was polypropylene [2]. But in 1996, poly-olefins and particularly polypropylene(PP) moved ahead of PET Fibers. They had 46% market shares in Fibers used for nonwovens, whereas, PET had a 45% share. By the end of 1998, olefin Fibers increased their share to 49% and PET dropped to 42%[25].
Mechanical properties of nonwoven fabrics depend on many parameters, including fiber properties, web structure and processing. It is, therefore, useful to review some of the elementary knowledge of fiber properties and other factors like web processing techniques and structure. What follows is a brief review of PET fiber properties, which should serve as background information for better understanding the subject.
POLYESTER Fibers
Polyester fiber is a " manufactured fiber in which the fiber forming substance is any long chain synthetic polymer composed at least 85% by weight of an ester of a dihydric alcohol (HOROH) and terephthalic acid (p-HOOC-C6H4COOH)" [3]. The most widely used polyester fiber is made from the linear polymer poly (ethylene terephtalate), and this polyester class is generally referred to simply as PET. High strength, high modulus, low _shrinkage, heat set stability, light fastness and chemical resistance account for the great versatility of PET.
-
POLYMER FORMATION
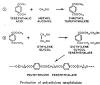
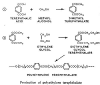
Polyethylene Teraphthalate (PET) is a condensation polymer and is industrially produced by either terephthalic acid or dimethyl terephthalate with ethylene glycol. [26] Other polyester Fibers of interest to the nonwovens field include:
( a ) Terephthalic Acid (PTA), produced directly from p-xylene with bromide-controlled oxidation.
( b ) Dimethyl Terephthalate (DMT), made in the early stages by esterification of terephthalic acid. However, a different process involving two oxidation and esterification stages now accounts for most DMT.
( c ) Ethylene Glycol ( EG ), initially generated as an intermediate product by oxidation of ethylene. Further ethylene glycol is obtained by reaction of ethylene oxide with water.
Equation 4 - PolyesterA
-
SYNTHESIS OF POLYMER
( 1 ) Linear Polyesters
A representative linear polyester, PET is polymerized by one of the following two ways: Ester Interchange: Monomers are dimethyl terephthalate and ethylene glycol. Direct Esterification: Monomers are terephthalic acid and ethylene glycol. Both ester interchange and direct esterification processes are combined with polycondensation steps either batch-wise or continuously. Batch-wise systems need two reaction vessels- one for esterification or ester interchange, the other for polymerization. Continuous systems need at least three vessels - one for esterification or shear interchange, another for reducing excess glycols, the other for polymerization.
Another way to produce PET is solid-phase polycondensation. In the process, a melt polycondensation is continued until the pre-polymer has an Intrinsic Viscosity of 1.0-1.4, at which point the polymer is cast into a solid firm. The pre-crystallization is carried out by heating (above 200oC) until the desirable molecular weight is obtained. Later the particulate polymer is melted for spinning. This process is not popular for textile PET Fibers but is used for some industrial Fibers.
( 2 ) Branched and Crosslinked Polyesters
If glycerol is allowed to react with a diacid or its anhydride each glycerol will generate one branch point. Such molecules can grow to very high molecular weight. If internal coupling occurs ( reaction of a hydroxyl group and an acid function from branches of the same or different molecule), the polymer will become crosslinked. Rigidly crosslinked polymers are totally unaffected by solvents.
3. fiber FORMATION
The sequences for production of PET Fibers and yarns depend on the different ways of polymerization (continuous, batch-wise, and solid-phase) and spinning (low or high windup speed) processes.
( 1 ) Spinning Process
The degree of polymerization of PET is controlled, depending on its end-uses. PET for industrial Fibers has a higher degree of polymerization, higher molecular weight and higher viscosity. The normal molecular weight range lies between 15,000 and 20,000. With the normal extrusion temperature (280-290oC), it has a low shear viscosity is 1000-3000 poise. Low molecular weight PET is spun at 265oC, whereas ultrahigh molecular weigh PET is spun at 300oC or above. The degree of orientation is generally proportional to the wind-up speeds in the spinning process. Theoretically, the maximum orientation along with increase in productivity is obtained at a wind-up speed of 10,000m/min. Although due to a voided skin, adverse effects may appear at wind-up speeds above 7000m/min.
( 2 ) Drawing Process
To produce uniform PET, the drawing process is carried out at temperature above the glass transition temperature (80-90oC). Since the drawing process gives additional orientation to products, the draw ratios (3:1-6:1) vary according to the final end-uses. For higher tenacities, the higher draw ratios are required. In addition to orientation, crystallinity may be developed during the drawing at the temperature range of 140-220oC. [26]
(3) Polyester fiber Flow Chart
Chart 1
(4) The latest Polyester production (Research Method)
Dr Boncella and Dr Wagner at The University of Florida are two scientists involved with the study to reveal a method for manufacturing polyester from two inexpensive gases: carbon monoxide and ethylene oxide.
The polyester most commonly used today is referred to as PET or polyethylene terepthalate. Scientists have been successful in producing low molecular weight polyester using carbon monoxide and ethylene oxide, but researchers still lack the catalyst - a substance that speeds up chemical reactions - needed to make the reaction work more efficiently. They are looking for the chemical compound that will take molecules of low DP and create 1arger ones. Although they have had success in the research so far, they have yet to produce a commercially useable polyester from the inexpensive gases. If this is successful, then these research findings can be used to replace the current polyester product, getting the same performance for a lower price. Finally, we all know that research requires patience and a long-term effort.[27]
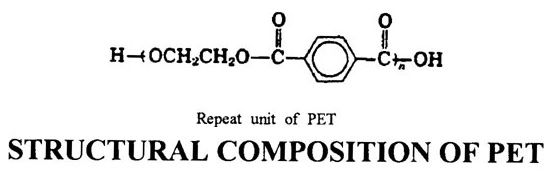
STRUCTURAL COMPOSITION OF PET
The one of the distinguishing characteristics of PET is attributed to the benzene rings in the polymer chain. The aromatic character leads to chain stiffness, preventing the deformation of disordered regions, which results in weak van der Waals interaction forces between chains. Due to this, PET is difficult to be crystallized. Polyester Fibers may be considered to be composed of crystalline, oriented semicrystalline and noncrystalline (amorphous) regions. The aromatic, carboxyl and aliphatic molecular groups are nearly planar in configuration and exist in a side-by-side arrangement. Stabilization distances between atoms in neighboring molecules are usually van der Waals contact distances, and there is no structural evidence of any abnormally strong forces among the molecules. The unusually high melting point of PET (compared to aliphatic polyesters) is not the result of any unusual intermolecular forces, but is attributed to ester linkages. The cohesion of PET chains is a result of hydrogen bonds and van der Waals interactions, caused by dipole interaction, induction and dispersion forces among the chains. The capacity to form useful Fibers and the tendency to crystallize depend on these forces of attraction.
The interactive forces create inflexible tight packing among macromolecules, showing high modulus, strength, and resistance to moisture, dyestuffs and solvents. The limited flexibility in the macromolecule is mainly due to the ethylene group. The extended quenched fiber does not show any early development of crystallinity; the growth of crystals starts to occur upon drawing. A number of basic structural models are required to represent the different states of the fiber: amorphous (no orientation) after extrusion, amorphous (no orientation) after cold drawing, crystalline orientation after thermal treatment and after hot drawing, stretching and annealing. The crystalline oriented form can also be obtained by high stress (high-speed) spinning.
Crystallinity and molecular orientation within the Fibers can be measured by Differential Scanning Calorimetry (DSC). This type of analysis is based on distinctly different values of the heats of fusion for crystalline and noncrystalline forms of the polymer. The heat of fusion of the sample is compared with a calibration standard. The crystallinity is determined by the following relationship
% Crystallinity = Equation 2
![]()
where ) Hf* is the heat of fusion of a 100% crystalline polymer, reported in the literature to be about 33.45 cal/g (equal to 140 J/g) [4]. The Tg (glass transition temperature) and Tm (melting point) of the Fibers can also be determined by DSC analysis. The results of the density and DSC measurements are shown in Table 1.
Table 1
| Density Gradient | DSC Measurements | |||||||||||
| fiber Type Base Fibers | Density (g/cc) | Crystallinity (%) | Tg1 (oC) | Tm2(oC) | ?? H3 (Cal/g) | Crystallinity (%) | ||||||
| A | 1.3803 | 41.22 | 154.3 | 251.3 | 17.19 | 51.38 | ||||||
| B | 1.3584 | 45.80 | 161.7 | 254.6 | 16.61 | 49.65 | ||||||
| C | 1.3809 | 41.73 | 152.9 | 255.8 | 15.29 | 45.73 | ||||||
| D | 1.3871 | 47.34 | 161.0 | 255.5 | 15.40 | 46.03 | ||||||
| E | 1.3825 | 43.71 | 175.9 | 257.4 | 16.41 | 49.05 | ||||||
1Glass transition temperature.
2Melting temperature.
3?H?--heat of fusion.
4Cold Crystallization range.
The rapid quenched PET without drawing is amorphous. The
temperature range of crystallization for PET is from 10oC below
the melting point to the temperature a little higher than the glass
transition temperature, 250-100oC. Typical PET has a 50%
crystallinity. The repeat unit of PET is 1.075 nm and is slightly shorter
than the length of a fully extended chain (1.09 nm). Therefore, the chains
are nearly planar. The crystal unit cell is triclinic with dimensions a =
0.456nm, b = 0.594nm, c = 1.075nm , ( = 98.5o,
= 118o
and ( = 112o.[11] PET crystal structure is illustrated in Fig. 1.
Another factor for crystallization is the position of the benzene rings. If
benzene rings are placed on the chain axis (c), then close packing of the
molecular chains eases polymer crystallization.
Figure 1
General Polyester fiber Characteristics:[24]
MELT-BLOWN PROCESS OF POLYESTER
The IV (intrinsic viscosity) and crystallinity levels of a melt-blown polyester determine the performance of the finished product. A higher IV leads to an increased level of crystallinity, which improves the barrier properties of the polyester melt-blown structure. However, it significantly reduces modulus, toughness and elongation. The advantage of using polyester over such polymers as polyolefins is its heat resistance and greater chemical resistance. Polyesters also offer a moderate oxygen barrier.
RELATIONSHIP BETWEEN STRUCTURE, PROPERTIES
AND PROCESSING PARAMETERS OF PET Fibers
Properties of polyester Fibers are strongly affected by fiber structure. The fiber structure, which has a strong influence on the applicability of the fiber, depends heavily on the process parameters of fiber formation such as spinning speed (threadline stress), hot drawing (stretching), stress relaxation and heat setting (stabilization) speed.
As the stress in the spinning threadline is increased by higher wind-up speed, the PET molecules are extended, resulting in better as-spun uniformity, lower elongation and higher strength, greater orientation and high crystallinity. Hot drawing accomplishes the same effect and allows even higher degrees of orientation and crystallinity. Relaxation is the releasing of strains and stresses of the extended molecules, which results in reduced shrinkage in drawn Fibers. Heat stabilization is the treatment to "set" the molecular structure, enabling the Fibers to resist further dimensional changes. Final fiber structure depends considerably on the temperature, rate of stretching, draw ratio (degree of stretch), relaxation ratio and heat setting condition. The crystalline and noncrystalline orientation and the percentage of crystallinity can be adjusted significantly in response to these process parameters.
Mechanical Properties
As the degree of fiber stretch is increased (yielding higher crystallinity and molecular orientation), so are properties such as tensile strength and initial Young's modulus. At the same time, ultimate extensibility, i.e., elongation, is usually reduced. An increase of molecular weight further increases the tensile properties, modulus, and elongation. Typical physical and mechanical properties of PET Fibers are given in Table 2. and stress-strain curves in Fig. 2. It can be seen that the filament represented by curve C has a much higher initial modulus than the regular tenacity staple shown in curve D. On the other hand, The latter exhibits a greater tenacity and elongation. High tenacity filament and staple (curve A and B) have very high breaking strengths and moduli, but relatively low elongations. Partially oriented yarn (POY) and spun filament yarns, exhibit low strength but very high elongation (curve E). When exposing PET fiber to repeated compression (for example, repeated bending), so-called kink bands start to form, finally resulting in breakage of the kink band into a crack. It has been shown in [5] that the compressibility stability of PET is superior to that of nylons.
Table 2
| Filment yarn | Staple and tow | |||||||
| Property | Regular tenacitya | High tenacityb | Regular tenacityc | High tenacityd | ||||
| breaking tenacity,e N/tex | 0.35-0.5 | 0.62-0.85 | 0.35-0.47 | 0.48-0.61 | ||||
| breaking elongation | 24-50 | 10-20 | 35-60 | 17-40 | ||||
| elastic recovery at 5% elongation, % | 88-93 | 90 | 75-85 | 75-85 | ||||
| initial modulus, N/texf | 6.6-8.8 | 10.2-10.6 | 2.2-3.5 | 4.0-4.9 | ||||
| specific gravity | 1.38 | 1.39 | 1.38 | 1.38 | ||||
| moisture regian, % | 0.4 | 0.4 | 0.4 | 0.4 | ||||
| melting temperature, oC | 258-263 | 258-263 | 258-263 | 258-263 | ||||
aTextile-filament yarns for woven and knit fabrics.
bTire cord and high strength, high modulus industrial yarns.
cRegular staple for 100% polyester fabrics, carpet yarn,
fiberfill, and blends with cellulosic blends or wool.
dHigh strength, high modulus staple for industrial
applications, sewing thread, and cellulosic blends.
eStandard measurements are conducted in air at 65% rh and 22oC.
fTo convert N/text to ge/den, multiply by 11.33.
gThe equilibrim moisure content of the Fibers at 21oC
and 65% rh.
Shrinkage varies with the mode of treatment. If relaxation of stress and strain in the oriented fiber is allowed to occur through shrinkage during fiber manufacture, then shrinkage at the textile processing stage is reduced and initial modulus is lowered. Polyester yarns held to a fixed length under tension during heat treatment are less affected with change in modulus, and reduced shrinkage values can still be obtained. This is very important in fiber stabilization.
PET shows nonlinear and time-dependent elastic behavior. It recovers well from stretch, compression, bending, and shear because of its relatively high initial modulus. Extensional creep occurs under load, with subsequent delay in recovery upon removal of the load. But compared with other melt-spun Fibers, the creep is small.
Figure 2
The formation of small fuzz balls of entangled Fibers (pills) on the fabric surface can be a serious problem. Fuzz formation may be affected by friction, stiffness, breaking strength and abrasion resistance. Shape, fineness, stiffness, recovery, friction and elongation influence entanglement of Fibers. After the pills have been formed, their rate of wear-off can affect the fabric appearance. Wear-off is a function of fiber breaking strength and flex life. Reducing the molecular weight which affects the abrasion resistance, flex life, and breaking strength, results in a decrease in pilling tendency of PET fiber. However, spinning low molecular weight linear PET fiber is difficult. As the molecular weight is reduced, the melt viscosity decreases and a uniform fiber with satisfactory continuity of spinning cannot be produced. Melt viscosity can be raised by the addition of a cross-linking compound, which is prone to hydroxyl groups. Another property, important especially to the apparel industry, is crimp stability or crimp compression. Generally, the tighter the packing of molecular chains, the stiffer and more mechanically resistant the fiber is. Crimp stability of the fiber can be improved with an increase in heating temperature. In addition, crimp compression of the fiber can be decreased by increasing draw ratio when the fiber is produced [6].
Chemical Properties
Polyester Fibers have good resistance to weak mineral acids, even at boiling temperature, and to most strong acids at room temperature, but are dissolved with partial decomposition by concentrated sulfuric acid. Hydrolysis is highly dependent on temperature. Thus conventional PET Fibers soaked in water at 70oC for several weeks do not show a measurable loss in strength, but after one week at 100oC, the strength is reduced by approximately 20%.
Polyesters are highly sensitive to bases such as sodium hydroxide and methylamine, which serve as catalysts in the hydrolysis reaction. Methylamine penetrates the structure initially through noncrystalline regions, causing the degradation of the ester linkages and, thereby, loss in physical properties. This susceptibility to alkaline attack is sometimes used to modify the fabric aesthetics during the finishing process. The porous structures produced on the fiber surface by this technique contribute to higher wettability and better wear properties [7].
Polyester displays excellent resistance to oxidizing agents, such as conventional textile bleaches, and is resistant to cleaning solvents and surfactants. Also, PET is insoluble in most solvents except for some polyhalogenated acetic acids and phenols. Concentrated solutions of benzoic acid and o-phenylphenol have a swelling effect.
PET is both hydrophobic and oleophilic. The hydrophobic nature imparts water repellency and rapid drying. But because of the oleophilic property, removal of oil stains is difficult. Under normal conditions, polyester Fibers have a low moisture regain of around 0.4%, which contributes to good electrical insulating properties even at high temperatures. The tensile properties of the wet fiber are similar to those of dry fiber. The low moisture content, however, can lead to static problems that affect fabric processing and soiling.
Optical Properties
PET has optical Figure 2 characteristics of many thermoplastics, providing bright, shiny effects desirable for some end uses, such as silk-like apparel. Recently developed polyester microfiber with a linear density of less than 1.0 denier per filament (dpf), achieves the feel and luster of natural silk [23].
Thermal Properties
The thermal properties of PET Fibers depend on the method of manufacture. The DTA (Fig. 3.) and TMA (Fig. 4) data for Fibers spun at different speeds show peaks corresponding to glass transition, crystallization, and melting regions. Their contours depend on the amorphous and crystalline content. The curves shown for 600 m/min and above are characteristic of drawn fiber. The glass transition range is usually in the range of 75oC; crystallization and melting ranges are around 130oC and 260oC, respectively.
Figure 3
The thermal degradation of PET proceeds by a molecular mechanism with random chain scission at ester linkages, although a radical mechanism has also been proposed. A chain-scission scheme is shown below:
The degradation products can undergo further changes, but at ordinary processing temperatures a certain proportion of carboxyl groups is introduced into the polymer structure. Color formation upon degradation has been attributed to the formation of polyenaldehydes from acetaldehyde and from a further breakdown of poly(vinyl ester)s.
DYEING PROPERTIES
Dyeing Properties
Because of its rigid structure, well-developed crystallinity and lack of reactive dyesites, PET absorbs very little dye in conventional dye systems. This is particularly true for the highly crystalline (highly drawn), high tenacity-high modulus Fibers. Polyester Fibers are therefore dyed almost exclusively with disperse dyes.
A considerable amount of research work has been done to improve the dyeability of PET Fibers. Polymerizing a third monomer, such as dimethyl ester, has successfully produced a cationic dyeable polyester fiber into the macro-molecular chain. This third monomer has introduced functional groups as the sites to which the cationic dyes can be attached [8]. The third monomer also contributes to disturbing the regularity of PET polymer chains, so as to make the structure of cationic dyeable polyester less compact than that of normal PET Fibers. The disturbed structure is good for the penetration of dyes into the fiber. The disadvantage of adding a third monomer is the decrease of the tensile strength.
A new dyeing process for polyester fiber at low temperature (40(C and below) has been reported [9]. This method employs a disperse dye in a microemulsion of a small proportion of alkyl halogen and phosphoglyceride. The main advantage of this method is low temperature processing, but there remains the environmental problem that is produced by using toxic carriers.
Another approach has been introduced by Saus et al [20]. The textile industry uses large amounts of water in dyeing processes emitting organic compounds into the environment. Due to this problem a dying process for polyester fiber was developed , in which supercritical CO2 is used as a transfer medium [21]. This gives an option avoiding water discharge. It is low in cost, non-toxic, non-flammable and recyclable. When dyed in an aqueous medium, reduction clearing is to be carried out to stabilize color intensity, which produces more waste water. Reduction clearing is not carried out following supercritical dyeing. Other advantages are better control of the dying process and better quality of application achieved.
Spunbond PET nonwoven webs have been treated by (SO2+O2) plasma and (N2+H2+He) plasma at the University of Tennesse, Knoxville. The research results show that spunbond PET nonwovens web can be colored by conventional water soluble acid dyes [10]. Plasma techniques open new avenues for coloring PET fabrics and are sure to be more evident in the coloring of polyester Fibers in the future.
Other Properties
Polyester Fibers display good resistance to sunlight but long-term degradation appears to be initiated by ultraviolet radiation. However, if protected from daylight by glass, PET fiber gives excellent performance, when enhanced by an UV stabilizer, in curtains and automobile interiors.Although PET is flammable, the fabric usually melts and drops away instead of spreading the flame. PET fiber will burn, however, in blends with cotton, which supports combustion.
Polyester has good oxidative and thermal resistance. Color forming species are produced and carboxyl end groups are increased. The resistance to both oxidative and thermal degradation may be improved by antioxidants. Mechanical properties are not affected by moderate doses of high-energy radiation. At doses of more than 0.5Mgy (Mrad), the tensile strength and ultimate elongation decrease, and deteriorate rapidly at 1-5 Mgy(100-500Mrad). Finally the resistance of polyester Fibers to mildew, aging and abrasion is excellent. Molds, mildew and fungus may grow on some of the lubricants or finishes, but do not attack the fiber.
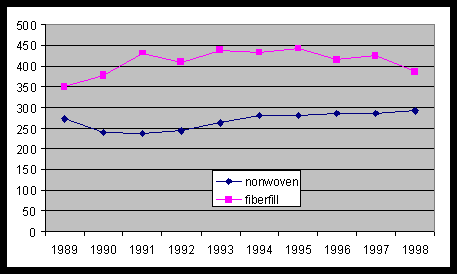
Consumption of PET Fibers for nonwovens/fiberfill(in million pounds)[25]
Graph
APPLICATIONS
The first U.S. commercial polyester fiber was produced by DuPont Company in 1953. Since polyester fiber has a lot of special characteristics, most of them are used in the following three major areas:[24]
|
Every form of clothing |
|
Carpets, curtains, draperies, sheets and pillow cases, wall coverings, and upholstery |
|
Hoses, power belting, ropes and nets, thread, tire cord, auto upholstery, sails, floppy disk liners, and fiberfill for various products including pillows and furniture |
Surgeon's gowns ,for example, were once woven linen but are now for the most part repellant treated entangled polyester fiber pulp composites on spunbond melt blown laminates. These new gowns are far superior to the older material in providing a breathable barrier between the surgeon and the patient, which serves to significantly reduce hospital infections. Spunlace mattress pad facing of 100% polyester continues to be the replacement of spunbond material because of the textile-like character of entangled fiber fabrics. PET has become the most important polymer type of fibrous prostheses. It is reasonably inert, bio-compatible, flexible, resilient and has an appropriate level of tissue acceptance. But, polymerization initiators, antioxidants, titanium dioxide and other impurities should be minimized to improve its bio-compatability.
Thermoplastics such as polyester are usually considered less flammable than cellulosic Fibers because they melt and shrink of drip away from the flame. Polyester resin such as Crystar, a DuPont trade name, is used to produce spunbonded polyester in a variety of applications: a nonwoven sheet fabric, fabric softener dryer sheets filtration media, apparel interlining, carpet backing, furniture and bedding, automotive seats and agricultural crop covers.
One of the important applications of PET is in the form of bicomponent Fibers. To increase the strength of the nonwoven fabric, in while maintaining the soft hand of LLDPE, PET is used in continuous bicomponent filaments having a sheath component made of LLDPE and a core component made of PET. The tensile strength of the fabrics is improved remarkably by the bicomponent filaments and depends on the LLDPE/pET ratio. The ultrasonically bonded polyester/polypropylene blend like Matarh's Ultraskin, the protective clothing, is said to protect wearers from rain while offering the breathability needed to provide comfort.
Dry and wet laid nonwovens made from a range of synthetic and inorganic Fibers are used in various insulation and industrial applications. A series of nonwoven polyester fiber mats are used in class F(155 c)DMD flexible electrical insulation laminates and electrical tape backing applications. Nonwoven mats made of polyester Fibers and high temperature resistant m-aramid are used as a cost effective replacement for aramid paper in class H(180 C) flexible electrical insulation composites.[28]
Composites made of 100% polyester Fibers are widely used as filtration media. Its layered structure gives excellent tear strength, a smooth, fiber free surface and edge stability. These products provide higher filtration efficiencies than spunbonded media that has not been calendered. The main advantage of these products is that they have no short Fibers to be carried downstream and contaminate the filtrate.[29]
In fiberfill applications polyester Fibers are used inside seat cushions, back pillows, mattresses and waterbeds, decorative and throw pillows, outdoor furniture and even hand-stuffed custom upholstery.[30]
REFERENCES
- Frohlich, Fritz W.: "Restructuring, Innovation See Akzo Nobel Through Difficult Business Environment ", International fiber Journal, (12), 3, 1997
- Harrison, David: "Synthetic Fibers for Nonwovens Update" Nonwovens Industry, 28 (6) 32- 39 (1997)
- Education Department, Man-made fiber Producers Association, Inc.: "Man-made fiber Fact Book", Page 20, (1978)
- Mehta, Aspy et al., "Equilibrium Melting Parameters of Poly (ethylene terephtalate)" J. of Polym. Sci., Polym. Phys. Ed. 16 (1978) 289
- Hearle, J.W.S., Miraftab, M.: "The Flex Fatigue of Polyamide and Polyester Fibers. Part II : The Development of Damage Under Standard Conditions" Journal of Materials Science 30(4) 1661-70 (1995)
- Pal, S. K., et al, "Draw-texturing of Microfiber Polyester yarn", Text. Res. J., 66(12) 770-776 (1996)
- Hsieh, Y.L. et al: "Wetting, Pore Structure, and Liquid Retention of Hydrolyzed Polyester fabrics "Text. Res. J., 66(1) 1-10 (1996)
- Pal, S. K., et al, "Draw-texturing of Cationic Dyeable Polyester yarn" Text. Res. J., 63(2) 71-79 (1993).
- Fite, G., F., J. et al.,: "Dyeing Polyester at Low Temperatures: Kinetics of Dyeing with Disperse Dyes", Text. Res. J, 65, (6), 362-368, (1995)
- Zhao, R., et al.: "Preliminary Research Report: Effects of Plasma Treatment on the Dyeability of PET Nonwoven Webs" , TANDEC document, University of Tennessee, Knoxville, 1997
- Lewin, M.,. Pearce, E. M .: fiber Chemistry: Handbook of fiber Science and Technology (IV), Marcel Dekker Inc., 1985.
- Moncrieff, R. W.: Man-made Fibers, 6th Ed., Newnes-Butterworths, 1975.
- Morton ,W. R., Hearle, J. W. S.: Physical Properties of Textiles Fibers, The Textile Institute, 1975.
- Peters, R. H.:Textile Chemistry: The Chemistry of Fibers, vol. I, Elsevier Publishing Co., 1963.
- Durso, D. F. et al: The Technical Needs: Nonwovens for Medical/surgical and Consumer Uses, TAPPI Press, Atlanta, GA, 1986.
- DuPont Magazine, 1997.
- Wood, Dennis E.: " Melt-blowing process for production of MicroFibers ", TANDEC Conference, University of Tennessee at Knoxville, 1991.
- Kubo, Eichi: "Bicomponent Spunbond fabric", TANDEC Conference, University of Tennesse, Knoxville, 1991.
- Acock, Harry R., Lampe, Frederick W.: "Contemporary Polymer Chemistry", Second Edition, Prentice Hall Inc., New Jersey, 1990.
- Saus, W., Krittel, D., Schollmeyer, E.: "Dyeing of Textiles in Supercritical CO2", Text. Res. J.,1993, 63, 135-142
- Saus, W., Krittel, D., Schollmeyer, E.: "Dyeing with Superficial CO2 An Alternative to High Temperature Dyeing of Polyester", Textile, 1992, 47, 1052-1054
- Harris, W.B.: "Is There a Future for Polyester Investments Outside Asia?", International fiber Journal, (11), 5, Oct.1996
- Fukuhara, Mototada: "Innovation in Polyester Fibers: from Silk-like to New Polyester", Text. Res.J. 63, July 1993, 387-91
- http://www.Fibersource.com/f-tutor/polyester.html
- David Harrison: "Shipments of Fibers to nonwovens reported for 1998", Nonwoven Industry, June 1999, 52
- J Gordon Cook: "Handbook of textile Fibers", Fourth Edition, 1968, P358 and P361
- http://www.eurekalert.org/releases/uf-cscmripc.html
- http://www.hollingsworth-vose.com/hovotherm.html
- http://www.reemay.com/filtration/fp_synergex.html
- http://www.dupont.com/fiberfill/dacron-main.html
Apparel Search
Add Your Company Contact
Us About Us Advertise
News Letter Legal
Help
Copyright ©
1999-2023 Apparel Search Company. All Rights Reserved.
Buy Fashion
For The Holidays.